Analysis Of The Park Patriot "Sarin Lab" In The Moscow Region
West of Moscow is a theme park called Patriot Park. One interesting exhibit at this park is an alleged Sarin production laboratory, said to have been captured in Syria. But is this really a production rig for the nerve agent Sarin?
Photographs of the “Sarin Lab” courtesy of the Conflict Intelligence Team (CIT)

Sarin Production
Sarin is a complicated substance to make. It is, in fact, more complicated to make than many other nerve agents, and there are numerous production pathways to get from basic materials to Sarin.
All of these production methods lead to the production of a substance called methylphosphonyl dichloride, which I will abbreviate top DC. To produce Sarin, DC needs to be converted to another chemical called methylphosphonyl difluoride, or DF for short. Regardless of the many pathways that lead to DF, they all lead to one of two final steps to get to the production of Sarin molecules. For lack of a better terminology, I will refer to them as Method 1 and Method 2.
Method 1 combines DF with isopropyl alcohol (IPA). One molecule of DF combined with one molecule of isopropyl alcohol reacts to produce one molecule of Sarin, one molecule of hydrogen fluoride (HF), and heat. Both the residual HF (which is a dangerous acid) and the residual heat are serious issues.
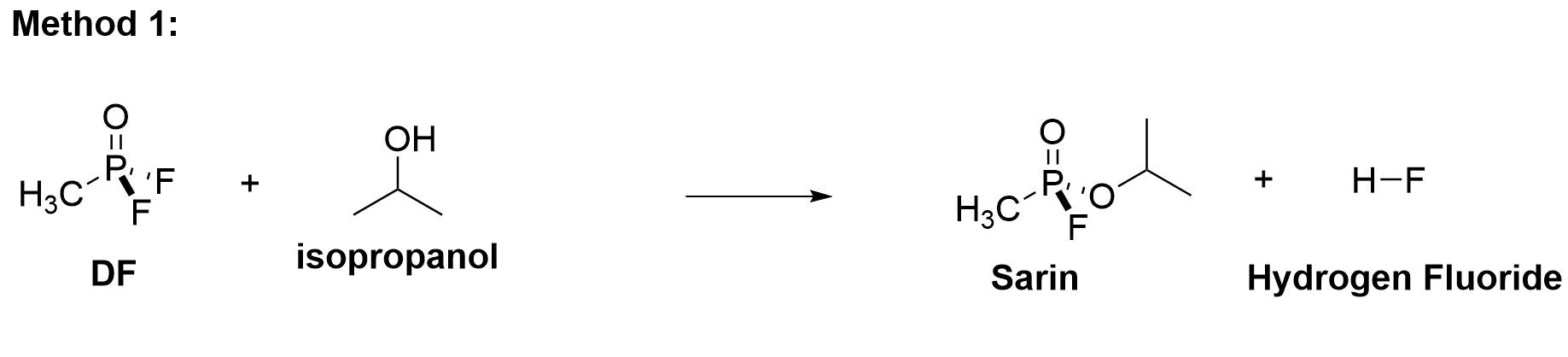
Method 2 was developed to cope with the problem of residual HF, which in large scale production proved to cause many difficulties, including deaths of production staff and loss of expensive hardware.
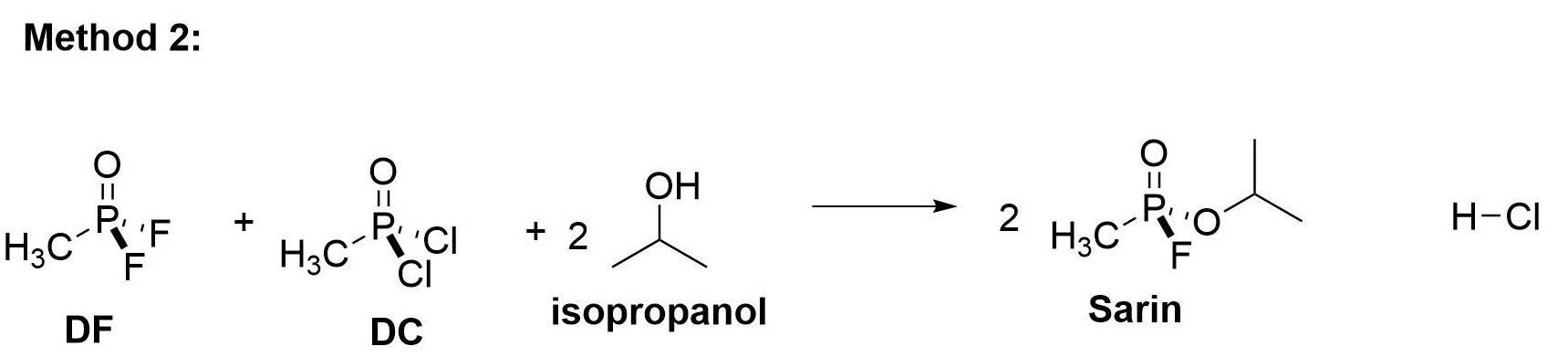
This method is more complicated. It combines a specialized mix of DF and DC with the IPA. This reaction combines 2 molecules IPA with 1 molecule of DC and 1 molecule of DF.
This reaction, when done carefully under the right conditions of temperature and pressure, yields 2 molecules of Sarin and 2 molecules of hydrogen chloride (HCl), as well as much heat. This residual HCl is easier to remove by various chemical engineering methods than residual HF, and is less dangerous to work with, but only in relative terms.
In the U.S. production method, this DC/DF cocktail was made by partially conducting the DC to DF conversion process and stopping the reaction when the appropriate ration of DC and DF was reached. This took literally years of trial and error to get exactly right at the U.S. Sarin production facility in Colorado.
The exact ratio of DC to DF is close to, but not exactly, equimolar. The timings, temperatures, and processes to convert DC to a DC/DF blend for this production method are still highly classified. It needs to happen under conditions of high temperature, because DC is a solid at room temperatures.
Unpacking The “Production Lab”
In overall terms, the displayed equipment rig is patently unsuitable for the production of Sarin. To anyone familiar with the well-documented historical processes for manufacture of Sarin in Germany, the U.S., and Japan, this equipment instantly looks inadequate to the task.
Indeed, it is hard to know where to start with this set-up.
There are a lot of things that are not explained in the exhibit display. Elements which are not present cannot, by definition, be evaluated — but their absence can be remarked upon. To be honest, it is quite possible that the persons who constructed this display did not know much about the process and were thus making guesses. There is no visible block diagram or process diagram purporting to show what is going on in this jumble of equipment.
If this is meant to be a Sarin production rig, it is showing only the final steps of the process, the last stages of Method 1 above.
There is no sufficient engineering or process controls for Method 2 to be contemplated. We are forced to assume that components are as marked, and that indicated chemical substances are as they are marked on their label. However, this may not actually be the case in the reality in the field. It is commonplace usage in clandestine drug laboratories to store substances in containers not consistent with their labelling.
What Is The Process On Display?
Which parts of the process are being done in which container is unclear. The blue drums are labelled “Reactor for mixing reagents” in English, but in Arabic the script translates to “reagents for reactor mixing” (note: I do not read Arabic and Bellingcat has provided this translation). These two descriptions are inconsistent. We are left with uncertainty as to whether one of the plastic drums is the reactor vessel or the metal cement mixer is the reactor.
It also is questionable why a covert lab would label its components. There are two main possibilities:
- DF is stored in one barrel and isopropyl alcohol is stored in the other. The two components are combined, along with triethylamine, in the cement mixer.
- The DF+IPA reaction is done in one of the plastic drums. The resulting product is then moved to the cement mixer where triethylamine is added.
Either way, there are many problems with these processes and this hardware. These are, in no particular order, described as follows:
The Absence of DF
You cannot make Sarin without DF, yet there is no evidence of any DF present.
The exhibitors have made the presence of isopropyl alcohol and the very optional triethylamine clearly visible, but not DF.
There is no container that looks adequate for storage of DF, which is highly reactive to many substances and even reacts with the ambient humidity in air. DF is not commercially available and cannot be obtained by purchase, as the only sources are closely regulated OPCW-inspected facilities that handle only very small quantities.
If you want DF, you have to make it. The various processes for making DF are complex and dangerous. But there is no indication of the necessary hardware or any process for making or purifying DF in these photographs. Anyone believing that this laboratory produces Sarin is required to assume that DF is already present. This, in turn, demands an explanation of where the DF came from.
The Blue Drums
The blue plastic drums are inherently incapable of handling the IPA-DF reaction.
Often, but not exclusively, produced from polyethylene, such large blue drums are widely used for storing chemicals. These particular drums are rated for storage of food. Their markings show that they are Russian in origin. They were procured by Russian company ZTI, near Moscow.
This raises an important issue. As such drums are ubiquitous around the world, why would a clandestine Syrian rebel group source them from a company in a town just west of Moscow? The drums came from a company only 60 km away from the exhibition site.

Accordingly to the manufacturer’s website, the drums appear to be made from either low density polyethylene (LDPE) or high density polyethylene (HDPE). This particular material has reasonable corrosion resistance.
Could a low density polyethylene drum be corrosion resistant enough for one or two production runs of Sarin? It is possible, although this material is not ideal for the purpose. The issue becomes one of heat and pressure. Polyethylene drums lose their structural integrity at higher temperature. They can withstand 80 degrees C, but can only stand 90 degrees C for a short period of time. The chemical compatibility of LDPE and HDPE is explained here.
Both the DF-IPA and DF-water reactions produce heat. Because the drums are pressurized, apparently with nitrogen gas or some other gas from the gas cylinders, the barrels will be under pressure.
It seems odd to pressurize a drum not designed for such use. The addition of heat and HF from the Sarin will greatly add to the pressure inside these barrels.
Such a drum will either burst or vent dangerous gases in a Sarin production run. It seems unwise and unsafe to use an LDPE or HDPE drum for the Sarin reaction. In addition, there is no visible mixing mechanism in the plastic drums. It is unclear how mixing could occur. 
The drums, on the other hand, could be suitable for storage of IPA and DF. DF is a difficult substance to store safely, as the U.S. military discovered during its own nerve agent manufacturing programme. DF’s preferred storage material is high density polyethylene (HDPE), a fact which is publicly published. The suitability of LDPE for storing DF is unclear in the available literature.
The Cement Mixer
Given the lack of mixing, the inconsistent labelling, and the fact that both of the plastic drums can’t both be used for the main DF-IPA reaction, the use of a modified cement mixer is extremely problematic for the DF-IPA reaction.
Anyone who has seen how a cement mixer such as this works knows that the mixer drum rotates. A sample video is here.
How are those improvised hose attachments going to work when the drum rotates? The input hoses are going to wrap around the mixer. They will break and spew chemicals everywhere.
Also, with the cover bolted shut, it is not clear how the end product is going to be removed. The metal mixer will get very hot very quickly.
There appears to be a vent on the left side. This will spew hot HF and Sarin in every direction while spinning. In addition, the HF and DF will attack the metal. Specialty metals are needed to contain a binary Sarin reaction for very long.
It is, on the other hand, possible that the intended use is not for the drum to rotate. This seems odd, as there is no other mixing mechanism visible or implied. It seems very likely that this mixer will fail, and fail catastrophically.

Connections And Fittings
Although the polyethylene drum will provide some corrosion resistance to the HF, the various fittings going into and out of the drum will not survive the HF or the DF. HF and DF are not kind to metal.
It should also be noted that the fittings on the gas cylinder have Cyrillic characters on them, indicating likely Russian origin. Why would a Syrian rebel chemical warfare effort source gas cylinder fittings from Russia, which would only highlight suspicion?

PPE
The protective equipment that the mannequins are wearing is not sufficient to safely handle the Sarin manufacturing process.
While the “Tyvek” suits and military filter-based protective mask are going to provide adequate protection against Sarin vapours and droplets, what they will not protect against is exposure to the Sarin components. Leaking HF vapour will most assuredly attack the skin of the person wearing that PPE, as the Tyvek is not an airtight suit.
Would the masks provide some protection to the respiratory tract? Likely. But will it protect someone for a protracted period of time while working in an enclosed space? Unlikely.
The gloves may not be adequate for triethylamine. It is not clear what type of glove is being showcased.

Temperature Controls
There is no hardware in evidence for controlling the temperature of any of the process. The heat produced in the reaction is going to do bad things if it isn’t controlled. The DF/IPA reaction is likely to produce enough heat to boil the contents of either the plastic barrel or the cement mixer. Either would lead to catastrophic failure. The sign on the exhibit claims that the process operates at 25 deg C. This is absurd.
Packaging And Labelling Of Chemicals
Neither of the containers appears to be the original container from the manufacturer. Safety information, in English, seems to be printed out and taped to the containers. We are left to assume that they are correctly labelled. But literally anything could have been in these containers.
The Alcohol
The pictured isopropyl alcohol does not appear to be the high purity required for safety and efficiency of the DF-IPA reaction.
For this reaction to work, it needs to be as close to 100% pure and completely free of moisture, particularly since DF reacts very badly with water. There is nothing shown in this production rig for purifying lower grades of IPA.
There’s no way that the pictured container contains 100% pure IPA, because the fluid level is clearly visible in the container. 100% pure IPA will rapidly absorb moisture out of ambient air. If you use this particular alcohol in the picture in a Sarin reaction, it is likely that there will be problems in the process. You will certainly produce Sarin, but quality and safety degrade quickly and you will need a more robust reaction vessel than the one illustrated.
DF reacts almost instantaneously to form HF and another acid. Any molecule of DF that reacts with water instead of IPA reduces the overall purity of the end product as that molecule is used up to make residue products instead of Sarin. Also, this water-DF reaction produces excess heat, which will add to the overall heat in the reaction vessel. In summary, use of the pictured alcohol makes the pictured reactor vessels inadequate.

Triethylamine
Various amine compounds have had a reasonable track record as acid reducing additives to Sarin. This topic has already been well explored on Bellingcat.
A thorough search of the technical literature will show that triethylamine is useful as a Sarin additive. It should be noted that this knowledge comes from the UK Sarin programme in the 1950s and is based on Sarin produced by Method 2.

It should be noted, however, that triethylamine’s safety is questionable in an improvised laboratory situation.
The physical characteristics of triethylamine make it a fire hazard. It is highly volatile and gives off vapours. It has a low flash point and a lower explosive limit that make it quite dangerous near any kind of heat source or spark. Given the possibility of a spark, increased temperatures due to exothermic reactions, and the likelihood of triethylamine vapours venting from the alleged mixing vessel, a fire or explosion is a distinct possibility. Also, triethylamine is corrosive to many types of plastics, rubbers, and metals.
Conclusion
Based on all available evidence, it is highly unlikely that this exhibit is actually a production laboratory for Sarin. While it may be fascinating for casual visitors to Patriot Park, it does not pass the authenticity test.
I acknowledge the help of several chemists, including DDTea in the preparation of this post. Thank you.
Bellingcat’s research for this publication was supported by PAX for Peace.




